
11 For example, a relatively large m value (i.e., a high susceptibility of the unfolding reaction to denaturant concentration) indicates that there is a large change in the exposure of apolar side chains on unfolding, which might be the case for a protein that has an extensive core of apolar side chains that are exposed upon denaturation. The m value also provides structural insights, as m values have been suggested to correlate with the change in solvent accessible apolar surface area upon unfolding of a protein. The Δ G o o, un value is a direct measure of the stability of a protein at the ambient solvent conditions, which can be moderate temperature and pH (e.g., 20 ☌ and pH 7). 10 Through an empirical relationship, the given equation appears to adequately describe the pattern for denaturant-induced unfolding of a number of proteins. 8 2.ĭenaturant-induced unfolding: The empirical relationship in Table I for chemical denaturation includes Δ G o o, un, the free energy change for unfolding in the absence of denaturant, and m, the denaturant susceptibility parameter (=–δΔ G un/δ), where is the molar concentration of added chemical denaturant. A consequence is that the Δ G o un( T) for unfolding of a protein will have a parabolic dependence on temperature and will show both high-temperature and low-temperature induced unfolding. That is, a positive Δ C p is a result of the hydrophobic effect. 6,7 The heat capacity change for unfolding of proteins is typically found to be positive and to be related to the increase in solvent exposure of apolar side chains upon unfolding. (7b) in Table I (the Gibbs-Helmholtz equation) should be used, where the Δ H o o,un and Δ S o o,un are values at some defined reference temperature, T o (e.g., 0° or 20 ☌). Both Δ H o un and Δ S o un may be temperature dependent, when the heat capacity change, Δ C p, has a nonzero value. Thermal unfolding: ΔH o un and Δ S o un are the enthalpy and entropy changes for a two-state unfolding reaction. We now present this derivation and encourage the reader to follow it through, and in the process, gain a deeper appreciation for the Second Law of Thermodynamics. 
This approach, which relies upon a fundamental mathematical theorem known as the Carathéodory theorem, is a most intellectually stimulating and satisfying exercise that leads to a much deeper understanding of the Second Law and the significance of entropy. However, it is possible to obtain equations (2.38) and (2.41) and show the equivalence of the thermodynamic and Absolute temperature scales without relying upon such idealized devices. Furthermore, in the next chapter we will apply the Carnot cycle while using an ideal gas as the working fluid to show that the thermodynamic (Kelvin) temperature scale and the ideal gas (Absolute) temperature scale are the same. In summary, we have seen how the introduction of the idealized Carnot engine leads to the definition of the thermodynamic temperature, an equation for calculating an entropy change from the flow of heat in a reversible process, and to the mathematical formulation of the Second Law in terms of entropy changes. Today, the Second Law, as applied to chemical systems, is firmly associated with the concept of entropy as expressed in the 1865 statement of Clausius and given mathematically by equation (2.41). The final volume of the gas V 2 is clearly greater than the initial volume V 1, so that ln(V 2/V 1 is a positive number. If U remains constant, so must the temperature thus T 2 = T 1. 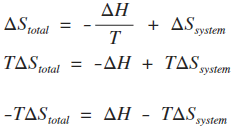
For an ideal gas U = mC vT the internal energy depends on T alone. It follows from the first law that dU = 0 the internal energy of the gas does not change. We therefore have dQ = 0 and dW = 0 at each stage of the expansion. The process is called a free expansion because of the absence of any external work as gas moves into the evacuated region. The rigid walls guarantee that no work is done on the system, that is, there can be no change in the volume of the system and thus no P dV work by the surroundings. 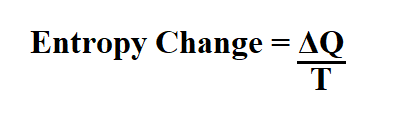
Thus, dQ = 0 for each stage of the expansion. Consider what happens when the partition ruptures, allowing the gas to expand and fill the entire container. Initially, one side contains an ideal gas and the other side is a vacuum. A vessel with rigid insulated walls is divided by a partition. Consider the experiment represented in Figure 24.11. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
